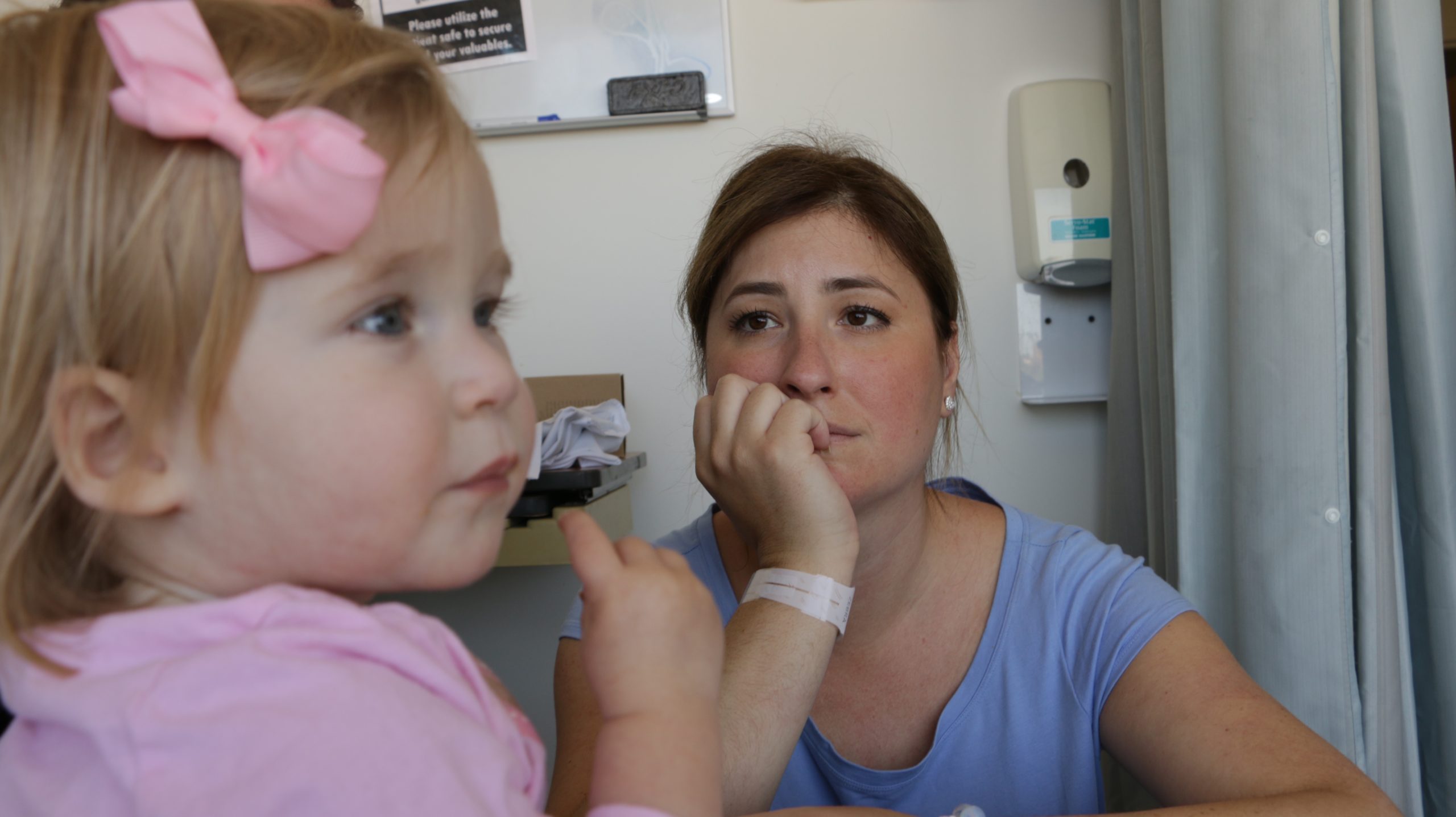
For the thousands of children and young adults worldwide who are afflicted with NPC and for the 60,000 children who have other neurodegenerative diseases, the APMRF’s goal means life. This research effort is a race against time. Medical research is costly, but it’s our hope in saving the lives NPC patients.
The Parseghian Fund currently funds multiple labs around the world. This funding supports an international research program:
- To expedite a treatment and cure for NPC disease
- To promote collaborative research efforts among the world’s leading experts in cholesterol metabolism, cell biology, neurobiology, genetics, pharmacology and other areas of science
- To study similar and further the cause of other neurodegenerative diseases as well as cholesterol storage disorders
The research program is directed by a Scientific Advisory Board (SAB) of internationally acclaimed scientists.

Current APMRF funded grants
APMRF is underwriting 18 projects for the 2023-24 Grant Funding Program. These support a collaborative group of leading Niemann-Pick Type C scientists from around the world.
Grants funded back a variety of high-impact investigations that include understanding the NPC pathway, the structure of the NPC1 protein, and how NPC proteins traffic cholesterol in and out of the cell.
Clinical Trial Patient Travel Reimbursement
The Parseghian Fund has set up a program to support the families transitioning from the Adrabetadex Clinical trial to an Expanded Access Program (EAP). Families can apply for awards to support the costs of travel.
Applying for Grants
The Ara Parseghian Medical Research Fund (APMRF) at Notre Dame (ND) is pleased to announce grant requests are now being accepted for the 2024 grant cycle. The mission of the APMRF is to promote research that furthers understanding of the molecular basis of Niemann Pick Type C (NPC) disease and targets development of novel strategies to treat and/or cure the disease.
More information on the grant process:
- Background and Purpose
The purpose of the Ara Parseghian Medical Research Fund (APMRF) is to promote research that is targeted toward understanding the molecular basis of Niemann Pick Type C (NPC) disease and developing novel therapeutic strategies to treat and/or cure the disease. In addition, because NPC disease is a cholesterol storage disease, it is anticipated that these studies should provide fundamental insight into cholesterol homeostasis and may serve to increase understanding of atherosclerosis and other cholesterol-related disorders. As part of our mission, the foundation will also seek to foster open collaboration between investigators currently working on NPC disease-related projects.The Scientific Advisory Board (SAB) of the APMRF is responsible for formulating the direction of research related to NPC disease. As such, the SAB will generate a series of scientific priorities and set forth specific research funding announcements (RFA’s). Investigators are encouraged to respond directly to these RFA’s. However, novel applications in NPC-related topics will also be considered under exceptional circumstances. Grant applications will be reviewed by the SAB via an expedited review mechanism. All funded projects will be reviewed by the SAB on an annual basis.
- Authority For Making Grants
Grants are made by the APMRF selection committee acting on the recommendations of the SAB. APMRF reserves the right to cancel a grant for cause at any time and require the return of any unexpended funds. - Eligibility For Grants
Grants are awarded to qualified institutions and organizations within the United States and abroad. - Duration Of Awards
Research grant awards are for a maximum of one year, but some proposals will be considered for a two year period. Overhead costs are limited to 10%. Investigators are encouraged to apply to other sources of support after the initial one funding period; however, they may also reapply for continued funding for either their current or a new research project. - Award Contract
Any approved awards will only be granted upon the signing of a grant award letter with the APMRF at ND. Please go to this link to review a sample award letter template. These include language regarding 1) the purpose of the grant, 2) use of grant funds, 3) subgrants and subcontracts, 4) reports and expectations, 5) report format, 6) record maintenance and inspection, 7) compliance, 8) indemnification, 9) publications, 10) intellectual property, 11) grant announcements, public reports and use of university name and logo, 12) equipment, 13) counterparts; original, 14) assignment; 15) independent contractors, 16) entire agreement, severability and amendment, 17) law and jurisdiction.